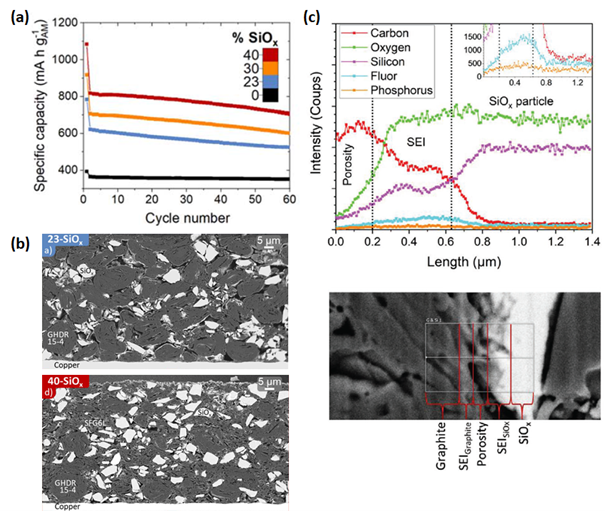
(a) Specific capacity vs. cycle number for Graphite/SiOx negative electrodes cycled in half-cells (vs. lithium metal), showing the increase in specific capacity with increasing SiOx content. (b) SEM observations of electrode cross section for 23 SiOx and 40 SiOx in backscattered electrons. (c) SEM-EDX elemental profiles obtained in the interface region between a SiOx and a graphite particle after 50 cycles for 23 SiOx, showing the presence of liquid electrolyte degradation compound forming the so-called solid electrolyte interphase (SEI). From C. Meyssonnier et al, Journal of The Electrochemical Society, 2024 171 070501 and C. Meyssonnier et al, Small Methods 2023, 2301370.
Electrochemical energy storage is an undisputed strategic challenge, but changing needs (mobile, stationary storage, etc.) and associated constraints (durability, resources, cost) mean that this is a constantly evolving field.
In our team of ten researchers with complementary expertise in chemistry, electrochemistry and physical chemistry, we explore different facets of electrochemical storage. Our work covers :
- synthesis and performance studies of innovative organic, inorganic and hybrid materials
- advanced formulation of electrodes, in particular silicon-based electrodes
- detailed analysis of operating and degradation mechanisms
- manufacture of pocket-size prototype cells (TRL 3)
- the development of functionalized coated current collectors.
Firmly rooted in the industrial ecosystem, we collaborate with key players in the sector: manufacturers of battery materials and components, battery assemblers, as well as major end-users such as automakers.
Applications: mobility and transport, energy storage, renewable energy storage, autonomous devices (connected objects, medical implants, etc.).
Keywords: halide, sulfides, operando characterizations, synchrotron and neutron diffractions, organic batteries, eco-compatible storage and organic or hybrid redox chemistry, sacrificial salts, zinc-ion battery, aqueous electrolyte, zinc electrodeposition, dendrites, hydrogen evolution, operando, Raman, EQCM-D, OEMS, FIB-SEM-Raman, coordination chemistry, MOFs, cross-linking, multi-technique analyses, spectroscopies, operando, energy, negative electrode (graphite, silicon, LTO), positive electrode (LFP, LMFP, NMC, LNMO), binder, electrode ink, electrode slurry, current collector, separator, recycling.
People involved: Bernard Lestriez and Philippe Poizot (theme coordinators), Nicolas Dupré, Joël Gaubicher, Philippe Moreau, Florent Boucher, Stéven Renault, Ivan Lucas, Patrick Soudan, Nicolas Goubard-Brétésché, Sarah Olivier-Archambaud
1 – Ceramic Electrolytes, Catholytes and Anolythes for All Solid-State Batteries
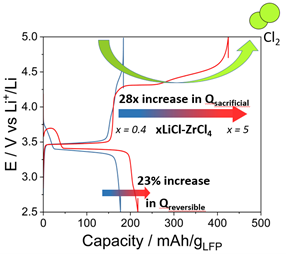
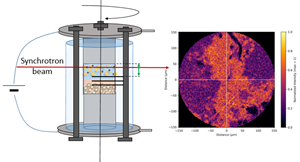
Figure 1a. Impact of the halide catholyte composition on the electrochemistry of LiFePO4 based cathodes
Figure 1b. Experimental setup and acquired data of operando Synchrotron-XRD CT experiment
Halide solid electrolytes, especially Li₂ZrCl₆ (LZC), represent a promising new class of materials for all-solid-state batteries (ASSBs). These electrolytes offer high oxidative stability (up to 4 V vs Li⁺/Li) and show partially reversible redox behavior, making them multifunctional components within the battery architecture. Researchers at IMN have discovered that halide catholytes can also act as secondary sources of electrons and lithium ions, boosting the energy output of ASSBs beyond the state-of-the-art. This innovative use, which led to a patent filed in March 2024, demonstrates the multifunctionality of halide compositions within the LixZrCl₄₊ₓ (LZC) family. These materials can act synergistically with conventional cathode materials to significantly increase cell-specific capacity, even at industrially relevant high cathode loadings (> 4 mAh/cm²). Furthermore, their irreversible redox capacity can be harnessed as a sacrificial agent for prelithiating micro-Si anodes or depositing lithium films in anode-less configurations. This “self-healing” approach paves the way for practical micro-Si and anode-free ASSBs. These findings are supported by advanced operando analyses such as Synchrotron X-ray Computed Tomography (SXCT), powder XRD, Mechanical Pressure Measurements (MCP), and Online Electrochemical Mass Spectrometry (OEMS), and highlight the synergy between halide catholytes and active materials like LMFP and LMO.
Expertise: Redox active Halide-based solid electrolytes, Operando characterization techniques (Synchrotron-XRDCT, Raman, Online Pressure), Sacrificial prelithiation in high areal capacity full cells
Keywords: Halide, Sulfides, operando characterizations, synchrotron and neutron diffractions
Collaborations
International: Prof. Shirley Meng, Chicago University, USA
France: Dr. Claire Villevieille, LEPMI (Grenoble) and Dr. Irina Profatilova, CEA-LITEN (Grenoble)
IMN Personnel Involved
Thematic Coordinators: Dr. Joël Gaubicher and Prof. Philippe Moreau
Doctoral Researcher: Mr. Branimir Stamenkovic, Adem Attafi
Master Student: Ms. Bétina Kaldé
Key Publications
B. Stamenkovic, Y. S. Meng, P. Moreau, and J. Gaubicher, J. Electrochem. Soc. 171, 050554 (2024). https://doi.org/10.1149/1945-7111/ad4b7f
B. Stamenkovic, E. Quarez, N. Dupré, P. Moreau, J. Gaubicher, ACS Materials Letters, 6, 4873-4880 (2024). https://pubs.acs.org/doi/abs/10.1021/acsmaterialslett.4c01497
2 – Organic Batteries, eco-friendly storage and organic redox chemistry
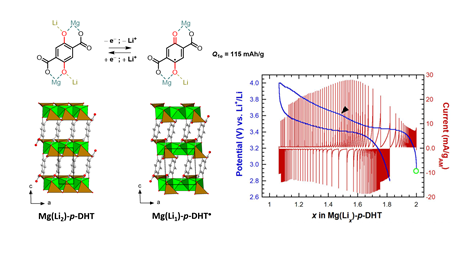
Figure: Asymmetric electrochemical lithium (de)insertion pathway in the organic host positive material in Magnesium (2,5-dilithium-oxy)-Terephthalate (Mg(Li2)-p-DHT), driven by a Peierls transition with spin-coupled dimer formation
To meet the challenges of sustainable development in the field of electrical energy management, eco-responsible technological solutions are urgently needed. This research theme focuses on the design, development and characterization of new organic and hybrid electrode materials for more environmentally-friendly electrochemical energy storage. In particular, the focus is on the development of innovative organic electroactive compounds that are potentially bio-sourced and capable of inserting both cations (n-type redox system) and anions (p-type redox system) into various electrolytic media. Our expertise in organic molecular engineering and electrochemistry (solid state and solution) also enables us to offer solutions for various battery technologies (sacrificial salts, electrocatalysis, etc.).
Expertise: Design, synthesis and characterization of organic and hybrid electrode materials, X-ray diffraction and in situ/operando measurements, organic redox systems.
Keywords: Organic batteries, eco-compatible storage and organic or hybrid redox chemistry, sacrificial salts
Collaborations :
National: Dr. Franck Dolhem (LG2A), Pr. Matthieu Becuwe (LRCS), Dr. Christine Frayret (LRCS), Dr. Thibaut Gutel (CEA Liten), Dr. Lionel Dubois (CEA Irig), Pr. Piétrick Hudhomme (Moltech Anjou)
International: Dr. Michel Armand (CIC EnergiGUNE), Prof. Daniel Brandell (Uppsala University), Prof. Birgit Esser (Ulm University), Dr. Alexandru Vlad (Université Catholique de Louvain)
Persons concerned: Philippe Poizot, Joël Gaubicher, Stéven Renault, Thomas Devic, Eric Quarez, Thierry Brousse
Current research projects: SONIC(Solid-state OrganicanioNIC battery, PEPR Batteries), ORGANION (Evaluation of rocking-chair ANIONic ORGanicbatteries for power, ANR-DFG), MUSIC(Materialsfor sUstainableSodium-Ion Capacitors, Europe)
Publications :
- A perspective on organic electrode materials and technologies for next generation batteries.
Birgit Esser, Franck Dolhem, Matthieu Becuwe, Philippe Poizot, Alexandru Vlad and Daniel Brandell.
Power Sources 2021, 482, 228814 - Opportunities and Challenges for Organic Electrodes in Electrochemical Energy Storage.
Philippe Poizot, Joel Gaubicher, Steven Renault, Lionel Dubois, Yanliang Liang and Yan Yao.
Rev. 2020, 120, 6490-6557 - Tuning the Chemistry of Organonitrogen Compounds for Promoting All-Organic Anionic Rechargeable Batteries.
Alia Jouhara, Eric Quarez, Franck Dolhem, Michel Armand, Nicolas Dupre and Philippe Poizot.
Chem. Int. Ed. 2019, 58, 15680-15684 - Raising the redox potential in carboxyphenolate-based positive organic materials via cation substitution. Alia Jouhara, Nicolas Dupre, Anne-Claire Gaillot, Dominique Guyomard, Franck Dolhem and Philippe Poizot.
Commun. 2018, 9, 4401 - Safe and recyclable lithium-ion capacitors using sacrificial organic lithium salt.
Jezowski, O. Crosnier, E. Deunf, P. Poizot, F. Beguin and T. Brousse.
Nat. Mater. 2018, 17, 167-173
3 – Zn-Ion Aqueous Batteries (ZIBs)
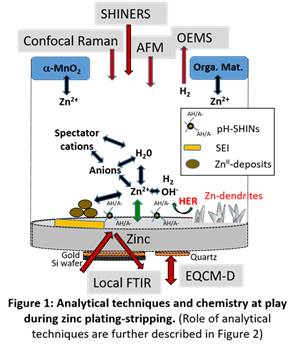
From an industrial perspective, a key benefit of Zn-MnO₂ technology is its inherent safety, due to the use of an aqueous electrolyte. Furthermore, the high theoretical mass capacities of zinc (820 mAh/g) and manganese dioxide (up to 616 mAh/g for the Mn⁴⁺/Mn²⁺ pair) make this an interesting electrochemical system. This has been exploited for many years in the production of non-rechargeable cells, such as saline and alkaline batteries. The current challenge is to develop a rechargeable version of the technology.
Within the IMN-led research effort, the main objective is to achieve a unified understanding of Zn/MnO2 dynamic interfacial chemistry and its morphological evolution across multiple scales. This is done using a unique combination of multi-scale operando characterization techniques, including enhanced Raman spectroscopy (SHINERS), Electrochemical Quartz Crystal Microbalance with Dissipation monitoring (EQCM-D), and Online Electrochemical Mass Spectrometry (OEMS).
This work also aims: (i) to extend this knowledge by studying the influence of the counter-electrode’s electroactive materials (Quinones, MnO2) and by designing novel electrolytic salts and functional additives, (ii) to identify, through collaboration with industrial partners, industrial-scale electrode or cell design that takes into account the constraints inherent to the MnO2 operation mechanism (successive electrodissolution and electrodeposition phenomena).
Expertises: Zinc electrodeposition and stripping, Zinc aqueous batteries (ZnB), Interfacial electrochemistry, Multi-scale operando characterizations (Raman, EQCM-D, OEMS), Electrolyte engineering and additive design, Morphological and structural diagnostics.
Keywords: zinc-ion battery, aqueous electrolyte, zinc electrodeposition, dendrites, hydrogen evolution, operando, Raman, EQCM-D, OEMS, FIB-SEM-Raman.
Collaborations:
- National: Dr. I. Profatilova (CEA Grenoble), Dr. C. Queffelec, Dr. Y. Pellegrin, Dr. F. Odobel (CEISAM, Nantes).
- International: Prof. M. Armand (CIC Energigune, Spain), Dr. E. Berg (Uppsala University, Sweden).
Industrial partner: Dr F. Chauveau (SAFT, Bordeaux France)
IMN Personnel Involved:
- Thematic Coordinator: Dr. J. Gaubicher. Participants: Prof. I. Lucas, Prof. P. Poizot, Prof. J. Barbe,
- Postdoctoral Researchers: Dr. Alissa Johnson, Dr. Arvinder Singh, D. Neau
Ongoing Research Projects:
- Unified understanding of zinc interfacial chemistry, Influence of organic electroactive materials on zinc behavior, Design of new salts and electrolyte additives (ANR ZORG – 2022-2026)
- Operando study of the MnO2/electrolyte interface in Zn-ion batteries (ANRT – SAFT/IMN – 2024-2027)
Key Publications: Singh et al. Electrochemistry Communications 154 (2023) 107559
4 – Coordination Polymers for Electrodes, Binders, and Coatings
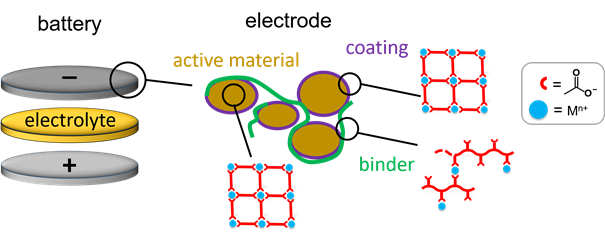
Coordination polymers (espescially the porous ones, also quoted Metal-Organic Frameworks, MOFs) were primarily investigated for their sorption/desorption properties, and found to be promising candidates for various applications including gas storage and capture (e.g.H2 andCO2, respectively), fluid separation, controlled drug release, and so on. More recently, this class of materials was also considered in the field of electrochemical energy storage, notably in secondary batteries such as metal-ion batteries. Electrodes of Li-ion batteries are complex, multiscale composite systems. Apart from the redox active material that stores and releases charges, other components of the electrodes which are present in a lower amount (conducting additive, binder, possibly coating) are also critical to achieve high performance. In the last years, we get interest in taking advantage of the characteristics of the coordination bonds (moderate strength, reversibility) and coordination networks (versatile composition, porosity, flexibility) to integrate such solids in electrodes for Li (or other alkali)-ion batteries either as redox active materials, coatings, or binders.
Expertises : Synthesis of materials, structural characterization, rheological studies, formulation of electrode, in situ and opendo studies
Keywords: chemistry coordination, MOFs, reticulation, multi-techniques analysis
Collaborations : Lionel Roué (INRS, Canada); Lorenzo Stievano, Long Nguyen, Nicolas Louvain, Fabrice Salles (ICG Montpellier)
Persons concerned: Bernard Lestriez, Thomas Devic
On-going projects : ANR : TEM-MOF, SIMPA, MOFSONG
Publications :
- Deciphering the benefits of coordinated binders in Si-based anodes by combined operando/in situ and ex situ X-ray micro- and nano-tomographies. V. Vanpeene, L. Huet, J. Villanova, M. Olbinado, F. Marone, E. Maire, L. Roué, T. Devic, B. Lestriez, Adv. Ener. Mater. 2024, 2403741.
- Fe(III)-carboxythiolate layered Metal-Organic Frameworks with potential interest as active materials for rechargeable alkali-ion batteries. N. Gedikoglu, P. Salcedo-Abraira, L. H. B. Nguyen, N. Guillou, N. Dupré, C. Payen, N. Louvain, L. Stievano, P. Poizot, T. Devic, J. Mater. Chem. A 2023, 11, 23909-23921.
- Is there any benefit of coating Si particles for negative electrode material for Li-ion batteries with a MOF? The case of Al fumarate. N. Kana, K. Touidjine, S. Olivier-Archambaud, E. Gkaniatsou, M. Paris, N. Dupré, N. Gautier, P. Moreau, C. Sicard, B. Lestriez, T. Devic, ACS Applied Energy Mater. 2023, 6, 9218-9230.
- Coordinatively cross-linked binders for silicon-based electrodes for Li-ion batteries: beneficial impact on mechanical properties and electrochemical performance. L. Huet, D. Mazouzi, P. Moreau, N. Dupré, M. Paris, S. Mittelette, D. Laurencin, T. Devic, L. Roué, B. Lestriez, ACS Applied Mater. Interfaces 2023, 15, 15509-15524.
- Nanoscale morphological characterization of coordinated binder and solid electrolyte interphase in silicon-based electrodes for Li-ion batteries. L. Huet, P. Moreau, N. Dupré, T. Devic, L. Roué, B. Lestriez, Small Methods 2022, 2200827.
- Deciphering the thermal and electrochemical behaviors of dual redox-active iron croconate violet coordination complexes. M. Denis, J.-M. Grenèche, N. Gautier, P. Poizot, T. Devic, Inorg. Chem. 2022, 61, 9308-9317.
5 – Development of advanced characterizations
RMN MAS
Multi-core MAS NMR spectroscopy is being developed and used at IMN to characterize the electrode/electrolyte interface and interfaces in the broadest sense. This is an innovative method, as it is still rarely used for this type of study. This approach, combined with other advanced characterization techniques such as XPS, EELS and SEM and TEM microscopy, has become a hallmark of the ST2E team in recent years. This approach is now routinely applied in various collaborative projects, increasingly involving industrial partners. NMR is also used to study lithiation mechanisms in positive and negative electrode materials, both inorganic and organic, and more recently to study the performance of solid electrolytes for lithium batteries.
Expertises: 1H, 6,7Li, 13C, 19F, 23Na, 31P, 35Cl, 51V, 93Nb NMR-MAS
Collaborations: M. Forsyth (Deakin University, Australia), C. Ling (Sydney University, Australia), B. Kang (Postech, South Korea), E. De Vito (CEA, Grenoble), J.N. Audinot (LIST, Luxembourg), L. Monconduit (ICGM, Montpellier), R. Berthelot (ICGM, Montpellier)
Person concerned: Nicolas Dupré
Current research project: ANR NanoLit
Publications :
- NMR Studies of Cathode Materials for Lithium-Ion Rechargeable Batteries, Dupré and C. P. Grey, Chem. Rev. 104(10) (2004) 4493
- Detection of Surface Layers Using 7Li MAS NMR, Dupré, J-F. Martin, D. Guyomard, A. Yamada, R. Kanno, J. Mat. Chem, 18 (2008) 4266
- Quantitative MAS NMR characterization of the LiMn1/2Ni1/2O2 electrode/electrolyte interphase, Cuisinier, J.F. Martin, P. Moreau, T. Epicier, R. Kanno, D. Guyomard, N. Dupre, Solid StateMagn. Res. 42 (2012) 51
- Interphase evolution of two promising Electrode materials for Li-ion batteries: LiFePO4 and LiNi1/2Mn1/2O2, Dupré, M. Cuisinier, J.-F. Martin, D. Guyomard, Chem. Phys. Chem. 15 (2014) 2
- Multi-probe study of the Solid Electrolyte Interphase on silicon based electrode in full cell configuration, Dupré, P. Moreau, E. De Vito, L. Quazuguel, M. Boniface, A. Bordes, C. Rudisch, P. Bayle-Guillemaud, D. Guyomard, Chem. Mat. 28(8) (2016) 2557
- Tuning the Formation and Structure of the Silicon Electrode/Ionic Liquid Electrolyte Interphase in Superconcentrated Ionic Liquids, Arano, S. Begic, F. Chen, D. Rakov, D. Mazouzi, N. Gautier, R. Kerr, B. Lestriez, J. Le Bideau, P. Howlett, D. Guyomard, M. Forsyth, N. Dupre, ACS Applied Materials & Interfaces, 13(24) 2021 28281
- Pristine surface of Ni-Rich layered transition metal oxides as a premise of surface reactivity, A.Laurita, L. Zhu, P.-E. Cabelguen, J. Auvergniot, J. Hamon, D. Guyomard, N. Dupré, P. Moreau, ACS Applied Materials & Interfaces (2022) 14 (37) 41945
- Lithium Diffusion-Efficient Ionogels as Polymer Solid Electrolyte for Next-Gen Lithium-Ion Batteries, Boluwatife, Y. Eddahani, P. Howlett, M. Forsyth, L. O’ Dell, N. Dupré, J. Le Bideau, Energy & Environmental Materials, 2024
Operan optical spectroscopies
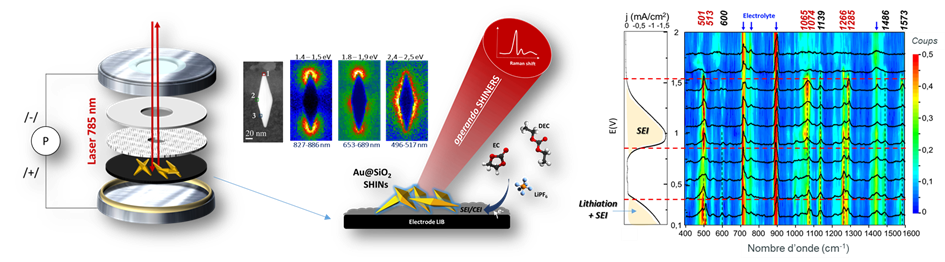
Operando SHINERS – Real-time monitoring of the composition of the electrode/electrolyte interface (< 100 nm) within operating batteries using exalted Raman spectroscopy (ERS). The electrode is pre-decorated with core-shell nanoparticles (SHINs), then assembled in a spectroelectrochemical cell of the button-cell type, representative of commercial batteries. The SHINs (here bipyramidal), illuminated by the Raman laser probe, act as local plasmonic amplifiers of the signal (near-IR plasmonic modes revealed by STEM-EELS), making it possible to extract the contribution to the signal of the fine interface between the electrode and the electrolyte, here a negative tin electrode in contact with the EC/DEC LiPF6 electrolyte.
Optical operando spectroscopy – A range of UV-Vis, fluorescence, Raman and infrared techniques are used at IMN to study energy systems and materials. Of particular interest are the richness of information in the chemical signatures produced, and the low-energy nature of the analysis (the visible or infrared light used is low-energy). Implementing these spectroscopic techniques under operando conditions is essential for understanding aging mechanisms, cycling processes and interactions between different electrode constituents, in order to optimize the performance of energy storage devices (LIBs, ZIBs, Fuel Cells). Optical spectroscopic methods developed at IMN under operando conditions include :
- MicroRaman spectroscopy for monitoring real-time compositional changes of electrode materials in contact with electrolytes during cycling
- SHINERS spectroscopy (derived from SERS surface-excited Raman spectroscopy) to decipher fine (electro)chemical processes at electrode/electrolyte interfaces (de-solvation, interphase formation, local pH)
- Fibered IR and Raman spectroscopy (compact, flexible probes under development) for analysis in complex, closed or sealed systems, or in harsh environments (high pressure & temperature, extreme pH)
- UV-Vis spectroscopy for monitoring redox properties and stability of organic materials under electrochemical conditions
- Fluorescence spectroscopy and electrochemical sensor for monitoring chemical cross-contamination processes in LIB batteries.
Expertise: optical spectroscopies, exalted spectroscopies (SERS, SHINERS, TERS), operando measurements, design of spectroelectrochemical cells, materials for electrochemical energy conversion and storage, electrochemistry.
Keywords: spectroscopies, operando, energy
Collaborations: SAFT, CEISAM, CIC (Spain) + European partners Opincharge….
People involved: Ivan LUCAS (ST2E), Joël GAUBICHER (ST2E), Bernard HUMBERT (PMN),
Current research projects :
- 2023-2026– European project OPINCHARGE (OPerando analyses and modelling of INterface dynamics and CHARGE transport in lithium-ion batteries) – Programme HORIZON-RIA call “Cross-sectoral solutions for the climate transition”, – 10 international partners (Luxembourg, Germany, Switzerland, Spain and France) – Leader: Santhana Eswara Moorthy – 5 million euros – IMN partners: P. Moreau and P. Abellan.
- 2024-2027: ARNT IMN/SAFT project – D. NEAU – “Etude operando de l’interface MnO2/électrolyte au sein des accumulateurs Zn-ion” – SAFT: F. CHAUVEAU, J. MARBAIX – Direction: I.T. LUCAS, co-direction J. GAUBICHER
- 2023-2027 – ANR Project ZORG – “Unified Understanding of the Zinc Aqueous (Electro)Chemistry from Multiscale Operando Investigations” – 42 months – 371 k€ – Project Leader: J. Gaubicher (IMN) – Partners Ceisam (F. Odobel, C. Queffélec et Y. Pellegrin), CEA (I. Profatilova, S. Genies), CIC (M. Armand).
- 2025-2028: Thesis ED3MG/Tandem Région – In situ/operando diagnosis by fibered vibrational spectroscopy – Direction I. LUCAS, co-direction B. Humbert
Publications :
- “Solid Electrolyte Interphase Instability in Operating Lithium-Ion Batteries Unraveled by Enhanced-Raman Spectroscopy,” ACS Energy Letters 6 (5), 2021 – 1757-1763 – https://doi.org/10.1021/acsenergylett.1c00436
- “Electrochemical tip-enhanced Raman spectroscopy for the elucidation of complex electrochemical reactions” Anal. Chem. 96, 2024, 2791-2798 – https://doi.org/10.1021/acs.analchem.3c02601″
- “Dynamics of transition metal dissolution and cross-contamination in operating Lithium-ion batteries,” Journal of Power Sources, 630, 2025, 236031 – https://doi.org/10.1016/j.jpowsour.2024.236031
STEM-EELS and STEM-EDX
Electron energy loss spectroscopy (EELS) in a transmission electron microscope (STEM-EELS) is developed and used in the team for general materials characterization, surface characterization and interface characterization. With the Nant’Themis, this is done on scales ranging from a few hundred nanometers to sub-nanometric scales. Oxidation degrees and compositions can be mapped ex situ at different cycling times. Technical developments are required to avoid sample degradation, a consequence of sample fragility and strong electron-matter interaction. This can be achieved by controlling the dose and type of probe sweep and/or making preferential use of low spectrum losses. STEM-EDX is also used for precise mapping, this time based on the measurement of X-rays emitted by the sample. This expertise is also used in collaboration with other IMN teams or other laboratories on various types of materials (piezoelectrics, MXenes, etc.).
Expertise: exploitation of low-loss spectra, monochromate spectroscopy, characterization of nanoscale interfaces, dose control for sensitive samples.
Collaborations: Thermo Fisher Scientific (Holland), Vincent Mauchamp (Pprime, Poitiers)
Person concerned: Philippe Moreau
Ongoing research projects: European OPINCHARGE project, ANR COMPAGNON
Publications :
- Nanoscale Morphological Characterization of Coordinated Binder and Solid Electrolyte Interphase in Silicon-Based Electrodes for Li-Ion Batteries, Lucas Huet, Philippe Moreau, Nicolas Dupré, Thomas Devic, Lionel Roué,* and Bernard Lestriez, Small Methods 2022, 2200827
- Pristine Surface of Ni-Rich Layered Transition Metal Oxides as a Premise of Surface Reactivity, Angelica Laurita, Liang Zhu, Pierre-Etienne Cabelguen, Jérémie Auvergniot, Jonathan Hamon, Dominique Guyomard, Nicolas Dupré, and Philippe Moreau ACS Appl. Mater. Interfaces (2022)
- Coordinatively Cross-Linked Binders for Silicon-Based Electrodes for Li-Ion Batteries: Beneficial Impact on Mechanical Properties and Electrochemical Performance, Lucas Huet, Driss Mazouzi, Philippe Moreau, Nicolas Dupré, Michael Paris, Sébastien Mittelette, Danielle Laurencin, Thomas Devic, Lionel Roué, and Bernard Lestriez, ACS Appl. Mater. Interfaces (2023)
- Structural and property engineering of 2D titanium carbides (MXene) thin films using ion irradiation, Ayoub Benmoumen, Marie-Laure David, Eric Gautron, Simon Hurand, Sophia Tangui, Lola Loupias, Aurélien Habrioux, Stéphane Célérier, Philippe Moreau, Vincent Mauchamp, Applied Surface Science 652 (2024) 159206
6 – Electrode formulation and prototyping of lithium-ion batteries
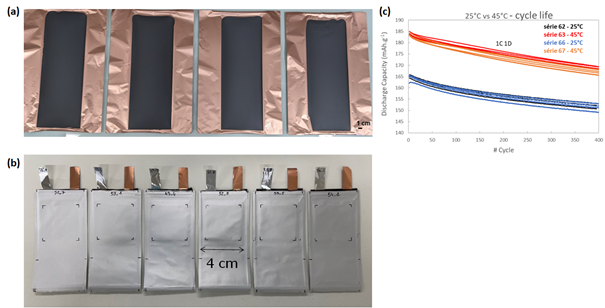
Figure. (a) Silicon-based negative electrode coatings on copper foil (A4 format) and (b) pocket cells prepared in the laboratory. (c) Cyclability of NMC811/Graphite pocket cells at 25 and 45°C (LP57+2%VC electrolyte) [2.7 – 4.2V].
Electrode formulation involves the selection of additives to the electroactive material powder, such as carbons to enhance electronic conductivity, and polymer binders to ensure mechanical cohesion and adhesion of the electrode film to the current collector throughout charge and discharge cycles. Electrode formulation also involves optimizing electrode manufacturing conditions, which involves optimizing electrode ink conditions, coating onto the current collector, drying and calendering. These last steps are essential for adjusting the porosity of the electrode, which in the battery is soaked in liquid electrolyte to ensure the electrode’s ionic conductivity.
We aim to understand the structuring mechanisms of electrode inks and the influence of polymer structure on electrode morphology, and the influence of electrode composition and morphology (i) on their mechanical and transport (electron and ion) properties; (ii) on their cycling durability; and (iii) on their ability to be charged and discharged more or less rapidly.
We are also studying the contributions of other components – separator, electrolyte and current collectors – to battery performance. We are working on commercial materials/components and those under development. Finally, we are looking into the direct recycling of battery materials and components.
Expertise: electrode formulation and manufacturing processes; polymer binders; assembly and cycling of button cells and pouch cells; morphology-properties-electrochemical performance relationships.
Keywords: negative electrode (graphite, silicon, LTO), positive electrode (LFP, LMFP, NMC, LNMO), binder, electrode ink, electrode slurry, current collector, separator, recycling.
Collaborations : Prof. Lionel ROUE (INRS, Canada), Dr. Eric Maire (MATEIS), Dr. Willy Porcher (CEA Liten)
People involved: Bernard Lestriez (Prof.), Nicolas Goubard-Bretesché (Research Engineer), Sarah Olivier Archambaud (Assistant Engineer), Aline Kneubl-Andreussi (PhD student), Natalie Herkendaal (PhD student), Cassandre Chalard (PhD student)
Current research projects: collaborative research contracts with companies (ARMOR, ARKEMA). PEPR batteries SONIC (Solid-state Organic anioNIC battery).
Publications :
- C. Meyssonnier, C. Chalard, A. Merabet, N. Goubard-Breteshé, N. Dupré, C. Paireau, B. Lestriez, “SiOx-rich electrode directly optimized using key formulation parameters”, J. Electrochem. Soc. 2024, 171, 070501, https://doi.org/10.1149/1945-7111/ad5a41
- J.A. Barreras-Uruchurtu, N. Besnard, C. Paul, L. Marchal, S. Devisme, B. Lestriez, “Effect of the physicochemical properties of PVdF on dry-sprayed graphite electrodes for lithium-ion batteries for electric vehicle applications”, J. Electrochem. Soc. 2024, 171, 12053, https://iopscience.iop.org/article/10.1149/1945-7111/ad9a08
- J. Xiong, N. Dupré, P. Moreau, B. Lestriez, “From the direct observation of a PAA based binder using STEM-VEELS to the ageing mechanism of silicon/graphite anode with high areal capacity cycled in an FEC-rich and EC-free electrolyte”, Adv. Energy Mater. 2022, 2103348, https://doi.org/10.1002/aenm.202103348
- M. Bichon, D. Sotta, E. De Vito, W. Porcher, B. Lestriez, “Performance and Ageing Behavior of Water-Processed NMC532/Graphite Lithium-Ion Cells”, J. Power Sources, 483, 2021, 229097, https://doi.org/10.1016/j.jpowsour.2020.229097
- S. J. Tambio, F. Cadious, E. Maire, N. Besnard, M. Deschamps, B. Lestriez, “The concept of effective porosity in the discharge rate performance of high-density positive electrodes for automotive application with the diffusion-limited penetration depth model”, J. Electrochem. Soc. 167, 2020, 160509, https://iopscience.iop.org/article/10.1149/1945-7111/abcb42
- V. Vanpeene, J. Villanova, A. King, B. Lestriez, E. Maire, L. Roué, “Dynamics of the morphological degradation of Si-based anodes for Li-ion batteries characterized by in-situ synchrotron X-ray tomography”, Adv. Energy Mater. 9, 2019, 1803947, https://doi.org/10.1002/aenm.201803947
- C. Reale Hernandez, A. Etiemble, D. Mazouzi, Z. Karkar*, E. Maire, D. Guyomard, B. Lestriez, L. Roué, “A Facile and Very Effective Method to Enhance the Mechanical Strength and the Cyclability of Si-Based Electrodes for Li-Ion Batteries”, Adv. Energy Mater, 2018, 8, 1701787, https://doi.org/10.1002/aenm.201701787
- C. Real Hernandez, Z. Karkar, D. Guyomard, B. Lestriez, L. Roué, “A film maturation process for improving the cycle life of Si-based anodes for Li-ion batteries”, Electrochem. Commun. 2015, 61, 102-105, http://dx.doi.org/10.1016/j.elecom.2015.10.014
- N. Delpuech, D. Mazouzi, N. Dupre, P. Moreau, M. Cerbelaud, J-S. Bridel, J-C. Badot, E. De Vito, D. Guyomard, B. Lestriez, B. Humbert. “Critical Role of Si Nanoparticles Surface on Lithium Cell Electrochemical Performance Analyzed by FTIR, Raman, EELS, XPS, NMR and BDS Spectroscopies”, J. Phys. Chem. C, 2014, 118, 17318-17331, https://pubs.acs.org/doi/10.1021/jp503949y
- P. N. Nguyen, N. A. Kumar, J. Gaubicher, F. Duclairoir, T. Brousse, O. Crosnier, L. Dubois, G. Bidan, D. Guyomard, B. Lestriez, “Nanosilicon-Based Thick Negative Composite Electrodes for Lithium Batteries with Graphene as Conductive Additive”, Adv. Energy Mater. 2013; 3; 1351-1357; https://advanced.onlinelibrary.wiley.com/doi/10.1002/aenm.201300330
- C. Fongy, A.C. Gaillot, S. Jouanneau, D. Guyomard, B. Lestriez, “Ionic vs Electronic Power Limitations and Analysis of the Fraction of Wired Grains in LiFePO4 Composite Electrodes”, J. Electrochem. Soc. 2010, 157, A885-A891, https://beta.iopscience.iop.org/article/10.1149/1.3432559
- W. Porcher, B. Lestriez, S. Jouanneau, D. Guyomard, “Design of Aqueous Processed Thick LiFePO4 Composite Electrodes for High-Energy Lithium Battery”, J. Electrochem. Soc. 2009, 156, A133-A134, https://iopscience.iop.org/article/10.1149/1.3046129/meta
- B. Lestriez, S. Bahri, I. Sandu, L. Roué, D. Guyomard, “On the binding mechanism of CMC in Si negative electrodes for Li-ion batteries”, Electrochem. Commun. 2007, 9, 2801-2806, https://doi.org/10.1016/j.elecom.2007.10.001
- D. Guy, B. Lestriez, D. Guyomard, “New architecture of composite electrode and improved battery performance from smart use of polymers and their properties”, Advanced Materials, 2004, 16, 553-557, https://advanced.onlinelibrary.wiley.com/doi/10.1002/adma.200306075
7 – Recycling of electrode materials
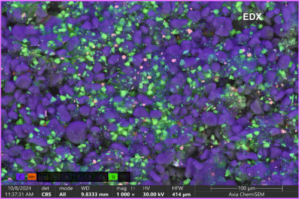
Figure. SEM-EDX image of recycled SiOx/Graphite powder with impurities (Manganese)
The growing use of Li-ion batteries, whether in portable devices or electric vehicles, makes resource and waste management crucial. Europe is introducing regulations to impose recycling and reuse quotas on battery manufacturers. The aim is to guarantee a flow of recycled raw materials and move towards a circular economy. As Europe has few (if any) deposits of nickel and other metals essential to battery manufacture, the exploitation of waste (urban mining) is becoming our main resource.
We work with partners specialized in recycling processes to evaluate and validate their processes. The questioning is on two levels:
- Is it possible to formulate electrode inks containing recycled materials that are stable and have the right rheology for the coating process? What is the acceptable impurity content according to their nature?
- Is it possible to assemble functional batteries from recycled materials, and what is their level of electrochemical performance (cyclability, power)? We need to understand the degree of regeneration of recycled material, and its performance compared with virgin material. The degree of regeneration can be defined at structural level (crystallinity, for example), morphological level (shape and granulometry), and compositional level (purity). The underlying issue is the trade-off between the cost/complexity of the material recycling process (separation, purification, regeneration), and the performance of the battery incorporating it.
Expertise: electrode formulation and manufacturing processes; polymer binders; assembly and cycling of button cells and pouch cells; morphology-properties-electrochemical performance relationships.
Keywords: recycling.
Collaborators: Dr. Jacob Olchowka and Dr. Gilles Philippot (ICMCB), Dr. Anna Vanderbruggen (Laboratoire GéoRessources, Université de Lorraine), Dr. Laure Monconduit and Moulay Sougrati (ICGM), Dr. Lenka Svecova (LEPMI)
People involved: Prof. Bernard Lestriez, Dr. Nicolas Goubard-Bretesché (Research Engineer), Sarah Olivier Archambaud (Assistant Engineer).
Ongoing research projects: PEPR recycling SYNERGIE (CloSed-loop recYcliNg of matERials from Graphite anodes and cathodes of Li-ion batterIEs).
Sub-themes
- 1 – Ceramic Electrolytes, Catholytes and Anolythes for All Solid-State Batteries
- 2 – Organic Batteries, eco-friendly storage and organic redox chemistry
- 3 – Zn-Ion Aqueous Batteries (ZIBs)
- 4 – Coordination Polymers for Electrodes, Binders, and Coatings
- 5 – Development of advanced characterizations
- 6 – Electrode formulation and prototyping of lithium-ion batteries
- 7 – Recycling of electrode materials