ANR project
Modeling Ionic-liquid Ionogel Interface Intercation In-silco
Dates:
From 01/01/2025 to 01/07/2028
Project coordinator:
Yann CLAVEAU (PMN team)
Partner laboratories:
IMN staff involved:
Jean LE BIDEAU (PMN), Chris EWELS (PMN), Samanvitha KUNIGAL (PMN)
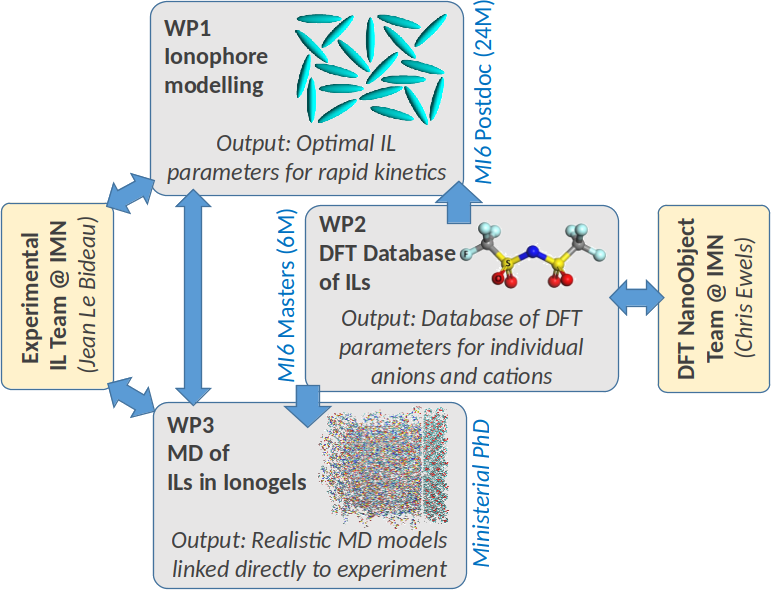
Modeling Interactions at the Interfaces of Ionic Liquids Ionogels
Ionic liquids (ILs) are salts that are liquid at room temperature. They are interesting candidates for applications in the energy field as safer electrolytes, as they have a higher energy storage density (batteries, supercapacitors). Their main drawback is their relatively high viscosity, due to the formation of aggregates that reduce their ionic conductivity. Our group has shown that confining an LI in a mesoporous host can counteract the formation of these aggregates and thus increase conductivity. However, the underlying mechanisms remain poorly understood and the experimental procedures difficult to optimize.
Unfortunately, these systems are very complex to simulate. To date, the literature has focused on molecular dynamics (MD) simulations of systems limited in size to a few cations and anions of interest. Generalization of the desired behavior, such as increased conductivity, has not been achieved. This is in direct contrast to a parallel field (liquid crystals) in which modeling has instead focused on interactions between idealized, simplified particle shapes.
The aim of MI6 is to open up a “rational design route” for selecting an ionic liquid and its confining host (ionogel), rather than using the trial-and-error approach currently followed by the community.
This approach will be employed with the use of an abstract structure model extended from the liquid crystal literature. It will optimize key LI properties for self-organization and fast ion transport. A database of DFT calculations of cations and anions will be specially created to select elements meeting the desired criterion. These candidates will then be tested using state-of-the-art MD models. This project will improve and optimize the conductivity of ionogels for energy storage applications.