ANR project
Ordered porous inorganic fluorides as efficient heterogeneous catalysts
Dates:
October 2020 – December 2024
Project coordinator:
Institut des Molécules et Matériaux (IMM Le Mans)
Partner laboratories :
- IMN
- Institut de Chimie des Milieux et Matériaux (IC2MP Poitiers)
- ROP SOLVAY Group / RHODIA OPERATIONS
IMN staff involved:
Chris EWELS
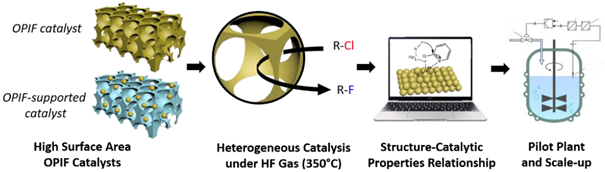
The element fluorine is used in fields as varied as medicine, energy, microelectronics and everyday plastics. Rare in its natural state, a considerable number of syntheses of fluorinated organic compounds have been developed using catalysts to improve both activity and selectivity. HF-catalyzed fluorination in the gas phase is widely applied on an industrial scale, particularly for non-functionalized aliphatic fluorinated compounds prepared from chlorinated precursors by Cl/F exchange. However, this strategy is not applicable to functionalized aliphatic fluorinated compounds, due to the sensitivity of most organic functions to HF, nor to aromatic fluorides, which are essentially produced by 2 liquid-phase reactions (Balz-Schiemann and HALEX). However, as these reactions are not very selective, they generate large volumes of effluent that cannot be recycled. It is therefore necessary to find new, more efficient, selective and environmentally-friendly fluorination methods. One such alternative, already successfully used for non-functionalized aliphatic fluorinated molecules, is the one-step fluorination of chlorinated aromatic molecules by C/F exchange under HF gas in the presence of catalysts. Besides being solvent-free, HCl is the only by-product that can be recycled. Recently, nanofluorides have been used as catalysts for the fluorination of 2-chloropyridine. While selectivity is optimal for this reaction, the significant activity, linked to the low Lewis acidity of the active sites, still needs to be improved by increasing the specific surface area of the catalyst. Indeed, given the severe operating conditions (HF gas at 350°C), nanofluorinated catalysts undergo a sintering process leading to a drastic loss of initially promising specific surface areas. This stumbling block forces us to explore innovative directions in order to develop materials that meet the 3 key requirements of a catalyst: activity linked to its specific surface, selectivity and stability, particularly under extreme operating conditions.
The innovation of the OPIFCat project is to prepare inorganic metal fluoride materials as efficient, selective and stable catalysts under the difficult conditions of fluorination of chlorinated reagents under HF. To this end, we will explore new architectures and innovative production methods based on ordered porous inorganic fluorides (OPIFs) designed to withstand such conditions, and whose development methodology will soon be patented by the IMMM team. The chemical composition of these OPIF catalysts will be guided by modeling the reactivity of the active sites. We will target new Cl/F exchange reactions involving aliphatic and aromatic nucleophilic substitution with five molecules applied to the fields of energy, agrochemistry and medicine. This project aims to understand the structure-activity relationship of the catalyst and to establish a “library of catalysts” with various Lewis acid strengths. The latter will help to rapidly select the most suitable catalyst for Cl/F exchange based on the characteristics of the reactant.
The OPIFCat project relies on a transdisciplinary consortium with complementary skills, and involves a major industrial group proactive in the sustainable energy transition. The consortium is made up of scientists with expertise in the development of fluorinated materials and polymers (IMMM) and in heterogeneous catalysis (IC2MP). It is completed by a specialist in modeling interactions in the presence of nanomaterials (IMN). Solvay will scale up the OPIF materials and validate their catalytic properties in a continuous tubular reactor.